Nicotinamide Riboside Chloride usage
A crystal form of NR chloride termed NIAGEN is generally recognized as safe (GRAS) for use in foods and the subject of two New Dietary Ingredient Notifications for use in dietary supplements. To evaluate the kinetics and dose-dependency of NR oral availability and safety in overweight, but otherwise healthy men and women, an 8-week randomized, double-blind, placebo-controlled clinical trial was conducted. Consumption of 100, 300 and 1000 mg NR dose-dependently and significantly increased whole blood NAD+ and other NAD+ metabolites within 2 weeks. The increases were maintained throughout the remainder of the study. There were no reports of flushing and no significant differences in adverse events between the NR and placebo-treated groups or between groups at different NR doses. NR also did not elevate low density lipoprotein cholesterol or dysregulate 1-carbon metabolism. Together these data support the development of a tolerable upper intake limit for NR based on human data.
All tissues produce NAD+ from NAM or the recently identified NAD+ precursor, nicotinamideriboside (NR). Some tissues can produce NAD+ from tryptophan de novo and nicotinic acid (NA), although the generation of NAD+ from tryptophan is much less efficient than from the vitamin precursors of NA, NAM, or NR, which are collectively termed vitamin B3. NAD+ can also be supported by dietary precursors. An important point to note is that, despite homeostatic systems and dietary intake of NAD+ precursors, it is now known that the levels of NAD+ co-enzymes are continuously challenged by metabolic stress. Although NA, NAM, and NR all produce NAD+ and NADP+, it is important to note that each precursor has unique effects physiologically.
NA:
NA can lower blood lipids and is used to treat dyslipidemia. At doses of greater than 50 mg/day, NA can also induce flushing.
NAM:
In contrast to NA, NAM does not lower blood lipids or cause flushing, has been reported be a sirtuin inhibitor at high doses, and appears to have a greater effect at elevating blood levels of homocysteine (HCY) in humans than NA via its metabolism to 1-methylnicotinamide (MeNAM).
NR:
In yeast, NR activates SIR2 and extends replicative lifespan. In mouse models, NR prevents high-fat diet-induced weight gain, fatty liver and diabetic peripheral neuropathy, noise-induced hearing loss, heart failure, and central brain injury. In addition, oral NR greatly improves survival and hematopoietic stem cell regeneration after irradiation of mice—an activity that was not seen in NA or NAM supplemented mice. In rats, oral NR promotes resistance to and reversal of chemotherapeutic neuropathy. In mice, oral NR increases the hepatic levels of the NAD+ metabolome with pharmacokinetics that are superior to that of NA and NAM. In addition, postpartum female mice and rats who were administered NR exhibited increased lactation and produced offspring that are stronger, less anxious, have better memory, and have enhanced adult hippocampal neurogenesis and body composition as adults. Because NR does not cause flushing or inhibit sirtuins and the genes (NRK1 and NRK2) required for the metabolism of NR to NAD+ are upregulated in conditions of metabolic stress, NR has a particularly strong potential as a distinct vitamin B3 like Magnesium L-Threonate Supplements to support human wellness during metabolic stress and aging.

 How Dietitians Play an Important Role in Helping People Stay Healthy?
How Dietitians Play an Important Role in Helping People Stay Healthy?  Finding the Right Health Care Provider: A Practical Guide to Choosing Quality Care
Finding the Right Health Care Provider: A Practical Guide to Choosing Quality Care  The Hidden Impact of Micro-Decisions: How Small Choices Shape Big Outcomes with insights from Joe Kiani, Masimo founder
The Hidden Impact of Micro-Decisions: How Small Choices Shape Big Outcomes with insights from Joe Kiani, Masimo founder  NATURAL AND MINDFUL WAYS TO MANAGE NICOTINE CRAVINGS
NATURAL AND MINDFUL WAYS TO MANAGE NICOTINE CRAVINGS  How Mind-Body Health Fits into Chronic Care
How Mind-Body Health Fits into Chronic Care  Gains of Using an App for Prediabetes
Gains of Using an App for Prediabetes  Treatment for Depression and Addiction: Why Both Need Care at the Same Time
Treatment for Depression and Addiction: Why Both Need Care at the Same Time 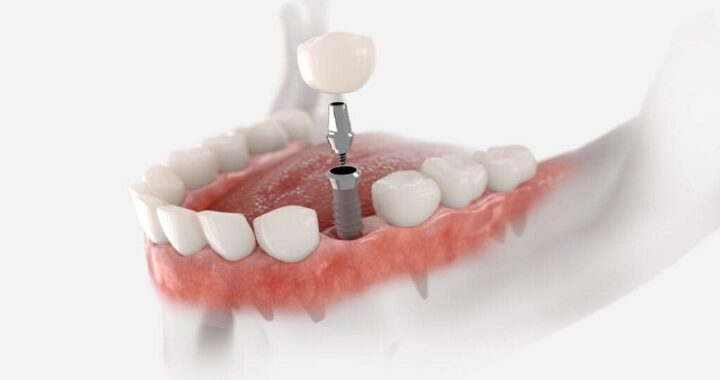 Dental Implant California: A Modern Solution for Restoring Missing Teeth
Dental Implant California: A Modern Solution for Restoring Missing Teeth